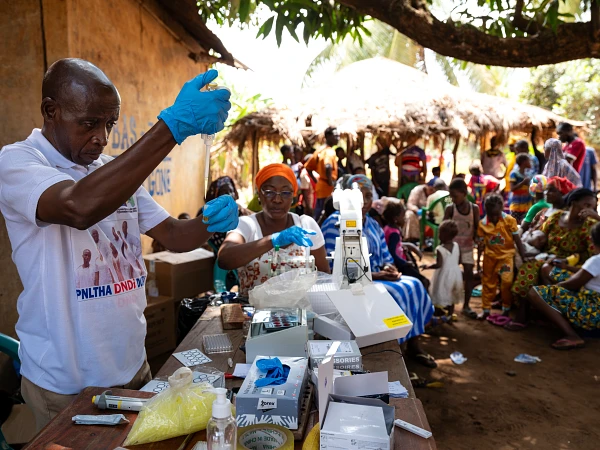
Acoziborole Winthrop receives greenlight as a three-tablet,single-dose treatment for most common form of sleeping sickness
Acoziborole will allow doctors to reach the most remote areas, where the last remaining pockets of the disease are. In 1998, nearly 40,000 cases were reported across Africa, and experts believe more than 300,000 others were never diagnosed.